 In the modern world, iron alloys, such as steel, stainless steel, cast iron and special steels, are by far the most common industrial metals, due to their mechanical properties and low cost. That event is considered the transition from the Bronze Age to the Iron Age. 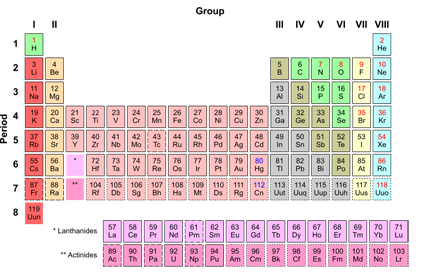
Humans started to master that process in Eurasia during the 2nd millennium BCE and the use of iron tools and weapons began to displace copper alloys-in some regions, only around 1200 BCE. Biological and pathological role Biochemistry Nutrition Diet Dietary recommendations Deficiency Excess ADHD Cancer Marine systemsĮxtracting usable metal from iron ores requires kilns or furnaces capable of reaching 1,500 ☌ (2,730 ☏) or higher, about 500 ☌ (932 ☏) higher than that required to smelt copper.Applications As structural material Mechanical properties Types of steels and alloys Catalysts and reagents Iron compounds.vanchor>:target~.vanchor-textProduction of metallic iron Laboratory routes Main industrial route Blast furnace processing Steelmaking Direct iron reduction Thermite process Molten oxide electrolysis History Development of iron metallurgy Meteoritic iron Wrought iron Cast iron Steel Foundations of modern chemistry. 
Chemistry and compounds Binary compounds Oxides and sulfides Halides Solution chemistry Coordination compounds Organometallic compounds Industrial uses. 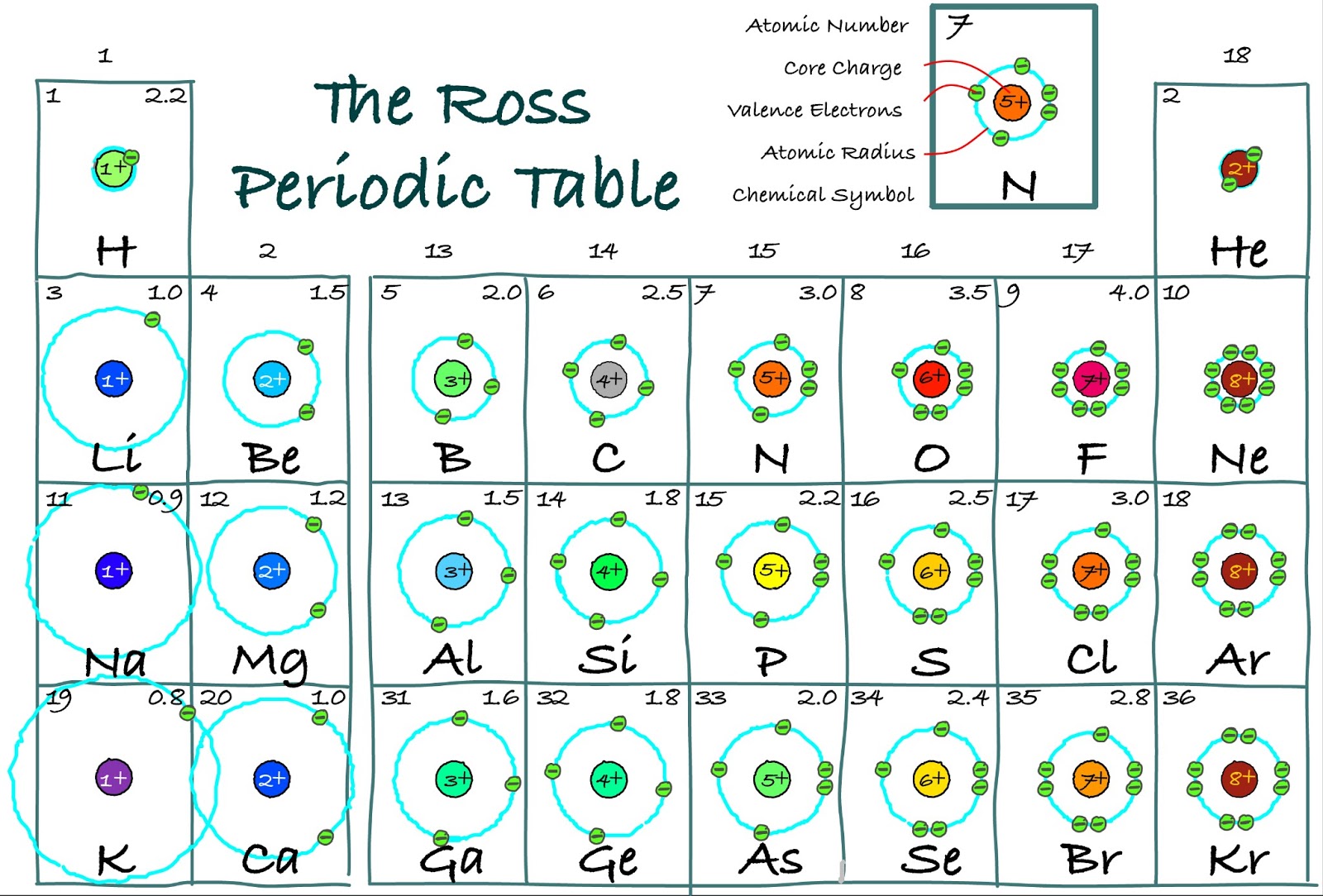
Origin and occurrence in nature Cosmogenesis Metallic iron Mantle minerals Earth's crust Oceans.Characteristics Allotropes Melting and boiling points Magnetic properties Isotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
